3.11 Spectroscopy and the Electromagnetic Spectrum
6 min read•january 2, 2023
Kanya Shah
Dalia Savy
Kanya Shah
Dalia Savy
What Is ?
Scientists began to understand and the way works by analyzing the that was emitted or absorbed by substances. 💡
The we see with our eyes👀, visible , is one type of ; because carries through space, it is also known as .
is carried in the form of , a type of quantum particle that acts as a "force-carrying particle" for electromagnetic energy. For example, when you turn on a flashlight🔦, trillions of shoot out, creating a beam of that illuminates a dark area. The same concept is applied in lasers and in many incredibly important measurements involving concentrations of solutions, as we'll see later in this unit when discussing the .
We'll get a deeper understanding of what really is and its role in the quantum world in the next study guide and key concept!
When we discuss , we typically refer to its and its . These two terms can help us describe how will act. is an interesting quantum idea because of the fact that it acts both as a particle (the photon) and as a wave. This is called . This goes back to what we discussed in unit one and electrons also exist as both particles and waves.
When thinking of a wave, it is useful to visualize it as a , oscillating back and forth periodically.
refers to the vertical height of the wave from the midline. The of a wave determines the 's intensity or brightness. The greater the , the brighter the .
(λ) refers to the length of one period of the wave. This can be described either as the peak-to-peak distance, or the zero-to-zero distance. It is typically measured in nanometers (nm), but it can also be measured in meters or micrometers. The of a wave determines the color of .
(ν) describes the number of waves that pass a fixed place in a given amount of time and is measured in cycles per second (s^-1) or Hertz (Hz). The hertz is defined as one cycle per second. The of a wave is directly proportional to the speed at which the wave is traveling. Basically, the faster the wave, the greater the .
and have an inverse relationship. Essentially, a high implies a small and vice versa. This relationship can be expressed through the equation c = λν, where c refers to the . We'll come back to this formula in the next section!
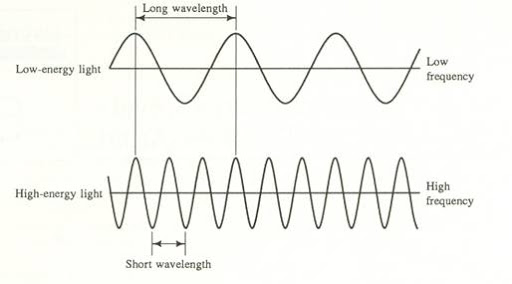
Image Courtesy of Florida State College
🌈
Visible , which is the we can see, is only a small category of . The electromagnetic spectrum includes all wavelengths of , ranging from very short to very long . A key trend to note is that can be characterized by its ; the shorter the , the higher the .
is the study of the interaction of and matter. diagram is shown below to use as a reference:
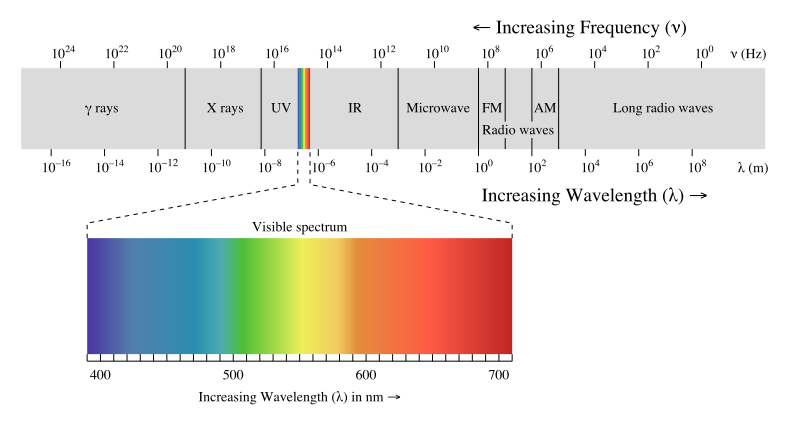
Image Courtesy of Khan Academy
There are a few things to notice about the electromagnetic (EM) spectrum, so let's break it down:
It is broken up into sections based on from ten-thousandths of nanometers long to hundreds of meters long. To put this in perspective, a nanometer is one billionth of a meter or 10^-9 meters.
If we start on the left of the EM spectrum, we can see gamma (γ) rays have the shortest and thus the highest . Because of their high frequencies, they can ionize radiation and are very dangerous. This means that they have enough energy to ionize atoms or molecules by removing their electrons and can easily pass through matter. This makes them very dangerous to humans and we are only exposed to them in small amounts in medical and industrial settings.
Next on the EM spectrum is , something you're already familiar with! have a longer and lower when compared to . Their properties allow them to pass through substances that block visible , which allows us to use them to image internal bones and organs!
Next, we got ultraviolet radiation or UV rays. Since their is still relatively high (although low compared to gamma and ), they are very harmful to us in high doses. UV rays are the reason why we have to wear sunscreen when exposed to sunlight for a prolonged period of time, as excessive exposure to UV increases the risk of skin cancer.
Taking a look at the EM spectrum, we can see that next is the , which is the EM radiation that we can see. The photoreceptors in our eyes are only receptive to within this spectrum, ranging from 400 nm in to 700 nm. Note that purple💜 has the shortest while red❤️ has the longest .
UV rays and visible are both associated with transitions in energy levels at an atomic level.
After the , we have infrared radiation, represented as IR on the EM spectrum above. All of the heat that we feel is infrared radiation since warm objects (even human bodies) emit at this ! IR is also associated with transitions in molecular vibrational levels.
Moving towards the opposite end of the spectrum, we are now at . are associated with transitions in molecular rotational levels. They are used in satellite systems and can cook food (hence our in the kitchen!).
How do microwave ovens warm our food? When you microwave food, the pass through the container and into the food. The are absorbed by the food, causing the molecules to vibrate and produce heat.
Last but not least, we have . have the longest and thus the lowest . Because of this, are not really dangerous to us. They are actually used to transmit signals responsible for radio, our cell signal, and television!
Here is another image that quickly summarizes the practical use of these different rays of .
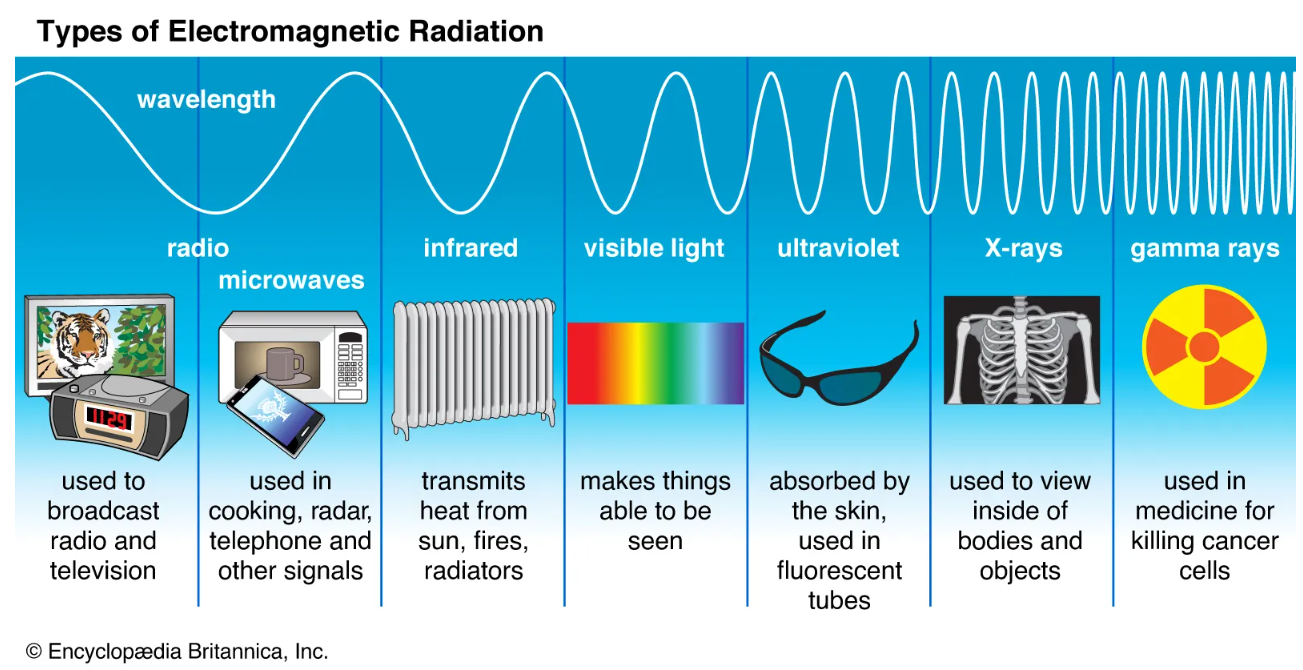
Image Courtesy of Britannica
Key Terms to Review (24)
3.11 Spectroscopy and the Electromagnetic Spectrum
6 min read•january 2, 2023
Kanya Shah
Dalia Savy
Kanya Shah
Dalia Savy
What Is ?
Scientists began to understand and the way works by analyzing the that was emitted or absorbed by substances. 💡
The we see with our eyes👀, visible , is one type of ; because carries through space, it is also known as .
is carried in the form of , a type of quantum particle that acts as a "force-carrying particle" for electromagnetic energy. For example, when you turn on a flashlight🔦, trillions of shoot out, creating a beam of that illuminates a dark area. The same concept is applied in lasers and in many incredibly important measurements involving concentrations of solutions, as we'll see later in this unit when discussing the .
We'll get a deeper understanding of what really is and its role in the quantum world in the next study guide and key concept!
When we discuss , we typically refer to its and its . These two terms can help us describe how will act. is an interesting quantum idea because of the fact that it acts both as a particle (the photon) and as a wave. This is called . This goes back to what we discussed in unit one and electrons also exist as both particles and waves.
When thinking of a wave, it is useful to visualize it as a , oscillating back and forth periodically.
refers to the vertical height of the wave from the midline. The of a wave determines the 's intensity or brightness. The greater the , the brighter the .
(λ) refers to the length of one period of the wave. This can be described either as the peak-to-peak distance, or the zero-to-zero distance. It is typically measured in nanometers (nm), but it can also be measured in meters or micrometers. The of a wave determines the color of .
(ν) describes the number of waves that pass a fixed place in a given amount of time and is measured in cycles per second (s^-1) or Hertz (Hz). The hertz is defined as one cycle per second. The of a wave is directly proportional to the speed at which the wave is traveling. Basically, the faster the wave, the greater the .
and have an inverse relationship. Essentially, a high implies a small and vice versa. This relationship can be expressed through the equation c = λν, where c refers to the . We'll come back to this formula in the next section!

Image Courtesy of Florida State College
🌈
Visible , which is the we can see, is only a small category of . The electromagnetic spectrum includes all wavelengths of , ranging from very short to very long . A key trend to note is that can be characterized by its ; the shorter the , the higher the .
is the study of the interaction of and matter. diagram is shown below to use as a reference:

Image Courtesy of Khan Academy
There are a few things to notice about the electromagnetic (EM) spectrum, so let's break it down:
It is broken up into sections based on from ten-thousandths of nanometers long to hundreds of meters long. To put this in perspective, a nanometer is one billionth of a meter or 10^-9 meters.
If we start on the left of the EM spectrum, we can see gamma (γ) rays have the shortest and thus the highest . Because of their high frequencies, they can ionize radiation and are very dangerous. This means that they have enough energy to ionize atoms or molecules by removing their electrons and can easily pass through matter. This makes them very dangerous to humans and we are only exposed to them in small amounts in medical and industrial settings.
Next on the EM spectrum is , something you're already familiar with! have a longer and lower when compared to . Their properties allow them to pass through substances that block visible , which allows us to use them to image internal bones and organs!
Next, we got ultraviolet radiation or UV rays. Since their is still relatively high (although low compared to gamma and ), they are very harmful to us in high doses. UV rays are the reason why we have to wear sunscreen when exposed to sunlight for a prolonged period of time, as excessive exposure to UV increases the risk of skin cancer.
Taking a look at the EM spectrum, we can see that next is the , which is the EM radiation that we can see. The photoreceptors in our eyes are only receptive to within this spectrum, ranging from 400 nm in to 700 nm. Note that purple💜 has the shortest while red❤️ has the longest .
UV rays and visible are both associated with transitions in energy levels at an atomic level.
After the , we have infrared radiation, represented as IR on the EM spectrum above. All of the heat that we feel is infrared radiation since warm objects (even human bodies) emit at this ! IR is also associated with transitions in molecular vibrational levels.
Moving towards the opposite end of the spectrum, we are now at . are associated with transitions in molecular rotational levels. They are used in satellite systems and can cook food (hence our in the kitchen!).
How do microwave ovens warm our food? When you microwave food, the pass through the container and into the food. The are absorbed by the food, causing the molecules to vibrate and produce heat.
Last but not least, we have . have the longest and thus the lowest . Because of this, are not really dangerous to us. They are actually used to transmit signals responsible for radio, our cell signal, and television!
Here is another image that quickly summarizes the practical use of these different rays of .

Image Courtesy of Britannica
Key Terms to Review (24)

