3.4 Electron configurations
1 min read•february 26, 2024
Understanding electron configurations is like getting a window into the world of atoms. Electrons are the key players in chemical bonding and reactions. In this study guide, we're going to dive deep into electron configurations and see how they influence everything from an element's placement in the periodic table to its role in technology.
⚛️ Electron Configuration Principles
The Aufbau Principle
The Aufbau principle states that electrons occupy the lowest energy orbitals available first. This rule helps us predict where electrons will be located around an atom's nucleus. Electron shells fill diagonally, from top to bottom and right to left, as demonstrated with the red arrows. The s orbitals can hold up to two electrons, the p orbitals hold up to 6 electrons, the d orbitals hold up to 10, and the f orbitals hold up to 14.
To illustrate, the Aufbau configuration for oxygen (which has 8 electrons) is .
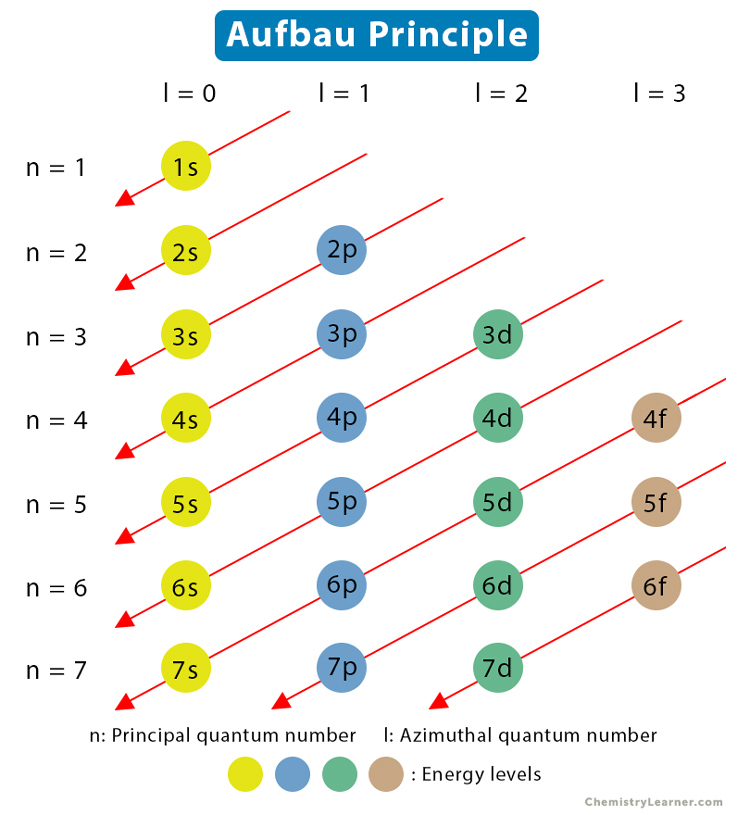
Image courtesy of Chemistry Learner
Solution:
Pauli Exclusion Principle
In the quantum mechanical understanding of atoms, electrons have something called spin. According to the Pauli exclusion principle, no two electrons in an atom can have identical quantum numbers; essentially, no two electrons can occupy the same state within an atom. Within any given orbital, you can only have two electrons, and they must have opposite spins. We represent the orbitals and spins with boxes and arrows, as demonstrated below.
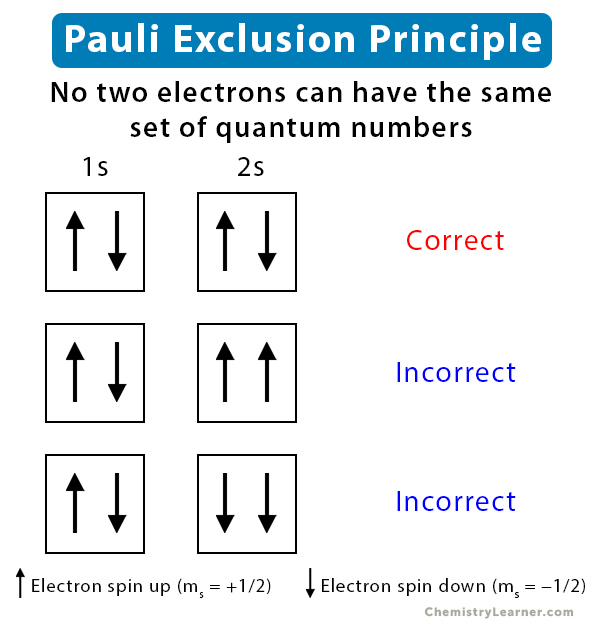
Image courtesy of Chemistry Learner
Hund's Rule
Hund's rule tells us that single electrons with the same spin must occupy each equal-energy orbital before additional electrons with opposite spins can join them.
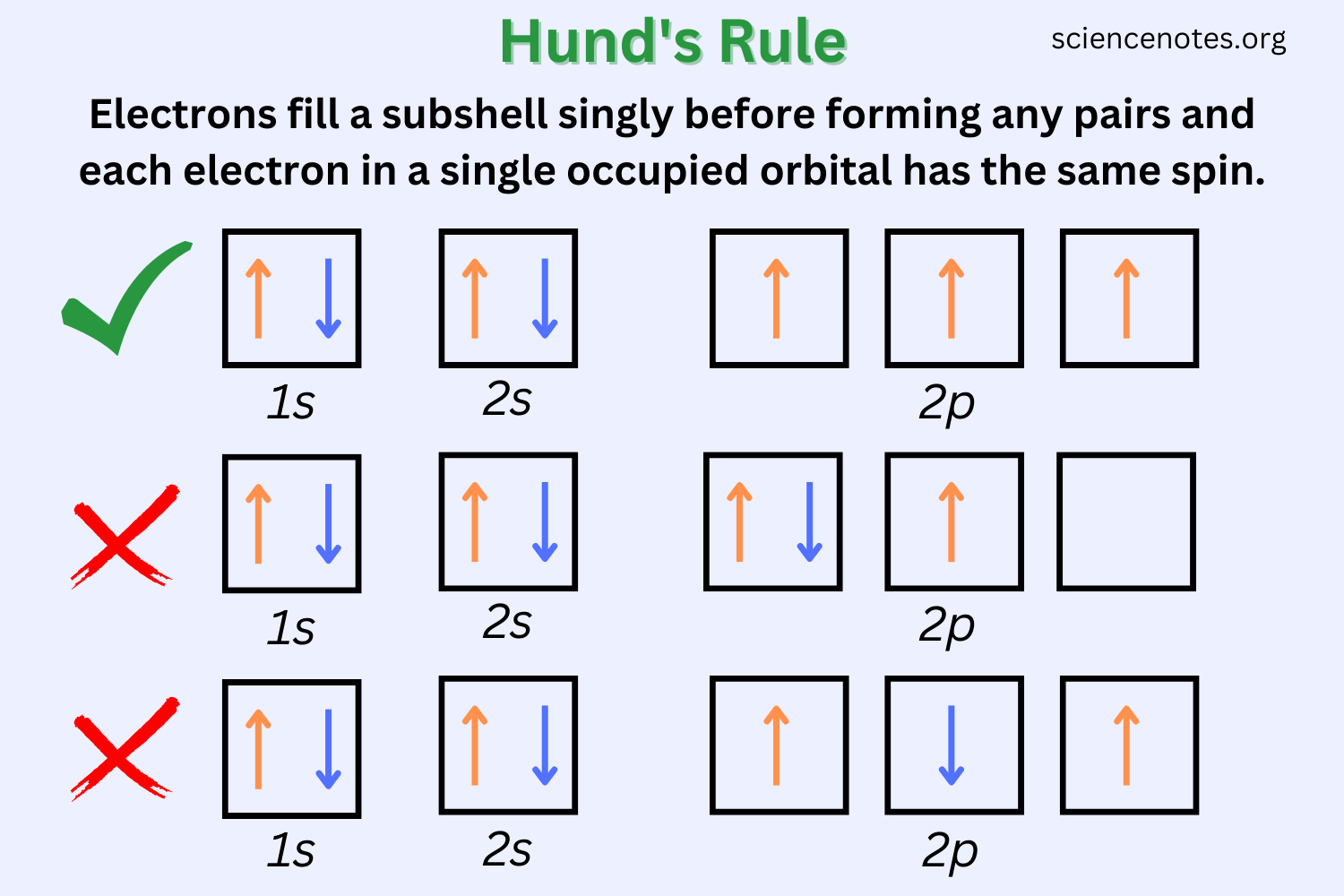
Image Courtesy of Science Notes
✏️ Applying It
🔢 Writing Electron Configurations
When writing electron configurations:
- Use the periodic table as your guide.
- Follow Aufbau’s principle.
- Remember that 's' orbitals hold 2 electrons, 'p' holds 6, 'd' holds 10, and 'f' holds 14.
- Write out the configuration using notation (e.g., ).
Practice Question: Write out the electron configuration for neon (Ne).
Answer:
💫 Orbital Diagrams
Orbital diagrams visually represent how electrons are distributed within an atom’s orbitals according to their energies and spin directions. These are the type of diagrams used when representing the Pauli exclusion principle and Hund’s rule. When drawing orbital diagrams:
- First find the Aufbau configuration.
- Draw the boxes for each orbital in the Aufbau configuration.
- Each box holds two electrons, so s orbitals get one box, p orbitals 3, d orbitals 5, and f orbitals 7.
- Begin filling the boxes with electrons, following both the Pauli exclusion principle and Hund’s rule.
Practice Question: Draw an orbital diagram for boron (B).
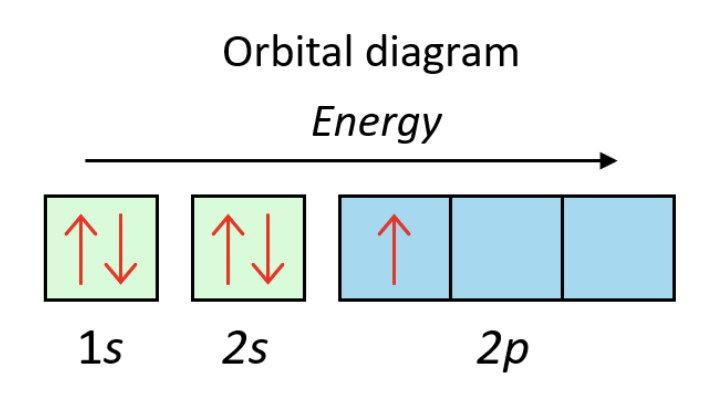
Image courtesy of Chemistry Steps
⛔ Exceptions
Certain elements like transition metals don't always follow expected patterns due to near-equal energy levels causing stability through half-filled or filled d-sublevels.
🍎 Applications
🐚 The Shell Model & Periodicity
The periodic table is arranged by increasing atomic number but also reflects repeating patterns of chemical properties which correlate with electron shell filling patterns.
- Period Trends: As you move across a period, each element has one more proton and one more valence electron than its predecessor.
- Group Similarities: Elements within the same group share similar valence shell configurations leading to similar chemical behaviors.
🔌 Valence Electrons & Chemical Reactivity
Valence electrons determine how elements bond and react since they are involved directly in forming chemical bonds.
🧪 Influence of Electron Configuration on Chemical Behavior
Electron arrangements affect reactivity:
- An atom may lose or gain electrons to achieve a full outer shell – think ions!
- Electronegativity relates to how strongly atoms attract shared electrons – noble gases are not electronegative because their shells are full.
- Metallicity: Metals tend to lose while nonmetals tend to gain or share.
🖇️ Bond Types & Electron Configuration:
- Ionic Bonds: Formed between metals and nonmetals; involve transfer of electrons.
- Covalent Bonds: Sharing of pairs of electrons between atoms.
- Metallic Bonds: Delocalized sea of shared free-flowing valence electrons among metal atoms contributing to conductivity and malleability.
🌐 Real World Implications
Knowledge of electron configurations extends beyond mere academic interest; it has practical applications:
- Predicting magnetic properties comes down to unpaired electron spins.
- Semiconductor behavior is based on specific electronic structures allowing movements under certain conditions.
- Understanding reactivity and bonding is crucial for synthesizing new drugs or materials with desired properties.
📌 Tips and Tricks
- Always start filling orbitals from lowest energy level up.
- Use mnemonic devices like "Smart People Don't Fret" (SPDF) order when remembering orbital types (
s,p,d,f), and don’t fret about your chemistry test! - Pay attention to exceptions especially for transition metals where traditional filling orders don’t always apply due to sublevel stability preferences.
- Relate everything back to real-world examples - this isn’t just theory!
Good luck! Remember that chemistry isn't about memorizing facts; it’s about understanding concepts so you can apply them creatively! 🧪🔬
3.4 Electron configurations
1 min read•february 26, 2024
Understanding electron configurations is like getting a window into the world of atoms. Electrons are the key players in chemical bonding and reactions. In this study guide, we're going to dive deep into electron configurations and see how they influence everything from an element's placement in the periodic table to its role in technology.
⚛️ Electron Configuration Principles
The Aufbau Principle
The Aufbau principle states that electrons occupy the lowest energy orbitals available first. This rule helps us predict where electrons will be located around an atom's nucleus. Electron shells fill diagonally, from top to bottom and right to left, as demonstrated with the red arrows. The s orbitals can hold up to two electrons, the p orbitals hold up to 6 electrons, the d orbitals hold up to 10, and the f orbitals hold up to 14.
To illustrate, the Aufbau configuration for oxygen (which has 8 electrons) is .

Image courtesy of Chemistry Learner
Solution:
Pauli Exclusion Principle
In the quantum mechanical understanding of atoms, electrons have something called spin. According to the Pauli exclusion principle, no two electrons in an atom can have identical quantum numbers; essentially, no two electrons can occupy the same state within an atom. Within any given orbital, you can only have two electrons, and they must have opposite spins. We represent the orbitals and spins with boxes and arrows, as demonstrated below.

Image courtesy of Chemistry Learner
Hund's Rule
Hund's rule tells us that single electrons with the same spin must occupy each equal-energy orbital before additional electrons with opposite spins can join them.

Image Courtesy of Science Notes
✏️ Applying It
🔢 Writing Electron Configurations
When writing electron configurations:
- Use the periodic table as your guide.
- Follow Aufbau’s principle.
- Remember that 's' orbitals hold 2 electrons, 'p' holds 6, 'd' holds 10, and 'f' holds 14.
- Write out the configuration using notation (e.g., ).
Practice Question: Write out the electron configuration for neon (Ne).
Answer:
💫 Orbital Diagrams
Orbital diagrams visually represent how electrons are distributed within an atom’s orbitals according to their energies and spin directions. These are the type of diagrams used when representing the Pauli exclusion principle and Hund’s rule. When drawing orbital diagrams:
- First find the Aufbau configuration.
- Draw the boxes for each orbital in the Aufbau configuration.
- Each box holds two electrons, so s orbitals get one box, p orbitals 3, d orbitals 5, and f orbitals 7.
- Begin filling the boxes with electrons, following both the Pauli exclusion principle and Hund’s rule.
Practice Question: Draw an orbital diagram for boron (B).

Image courtesy of Chemistry Steps
⛔ Exceptions
Certain elements like transition metals don't always follow expected patterns due to near-equal energy levels causing stability through half-filled or filled d-sublevels.
🍎 Applications
🐚 The Shell Model & Periodicity
The periodic table is arranged by increasing atomic number but also reflects repeating patterns of chemical properties which correlate with electron shell filling patterns.
- Period Trends: As you move across a period, each element has one more proton and one more valence electron than its predecessor.
- Group Similarities: Elements within the same group share similar valence shell configurations leading to similar chemical behaviors.
🔌 Valence Electrons & Chemical Reactivity
Valence electrons determine how elements bond and react since they are involved directly in forming chemical bonds.
🧪 Influence of Electron Configuration on Chemical Behavior
Electron arrangements affect reactivity:
- An atom may lose or gain electrons to achieve a full outer shell – think ions!
- Electronegativity relates to how strongly atoms attract shared electrons – noble gases are not electronegative because their shells are full.
- Metallicity: Metals tend to lose while nonmetals tend to gain or share.
🖇️ Bond Types & Electron Configuration:
- Ionic Bonds: Formed between metals and nonmetals; involve transfer of electrons.
- Covalent Bonds: Sharing of pairs of electrons between atoms.
- Metallic Bonds: Delocalized sea of shared free-flowing valence electrons among metal atoms contributing to conductivity and malleability.
🌐 Real World Implications
Knowledge of electron configurations extends beyond mere academic interest; it has practical applications:
- Predicting magnetic properties comes down to unpaired electron spins.
- Semiconductor behavior is based on specific electronic structures allowing movements under certain conditions.
- Understanding reactivity and bonding is crucial for synthesizing new drugs or materials with desired properties.
📌 Tips and Tricks
- Always start filling orbitals from lowest energy level up.
- Use mnemonic devices like "Smart People Don't Fret" (SPDF) order when remembering orbital types (
s,p,d,f), and don’t fret about your chemistry test! - Pay attention to exceptions especially for transition metals where traditional filling orders don’t always apply due to sublevel stability preferences.
- Relate everything back to real-world examples - this isn’t just theory!
Good luck! Remember that chemistry isn't about memorizing facts; it’s about understanding concepts so you can apply them creatively! 🧪🔬

Resources
© 2024 Fiveable Inc. All rights reserved.
AP® and SAT® are trademarks registered by the College Board, which is not affiliated with, and does not endorse this website.
