5.2 Understanding Molecular and Formula Weights
1 min read•february 27, 2024
Now that we got the concept of the mole familiarized with in the previous guide, a silly question you might have when first introduced to the idea that atoms and molecules have “weight” and “masses,” too, is: “How do we even weigh them?” 😳
Good news, you’re in the right place! This guide will help you unravel the mystery of how we quantify the mass of molecules and ionic compounds, which is necessary to do calculations in chemistry.
📐 Calculating Molecular and Formula Weights
From Chemical Formulas to Weights
We briefly touched upon this when looking at mole fundamentals, but now that we’re diving into molecular and formula weights, let’s break this process down into steps to make things easier for ourselves! 😁
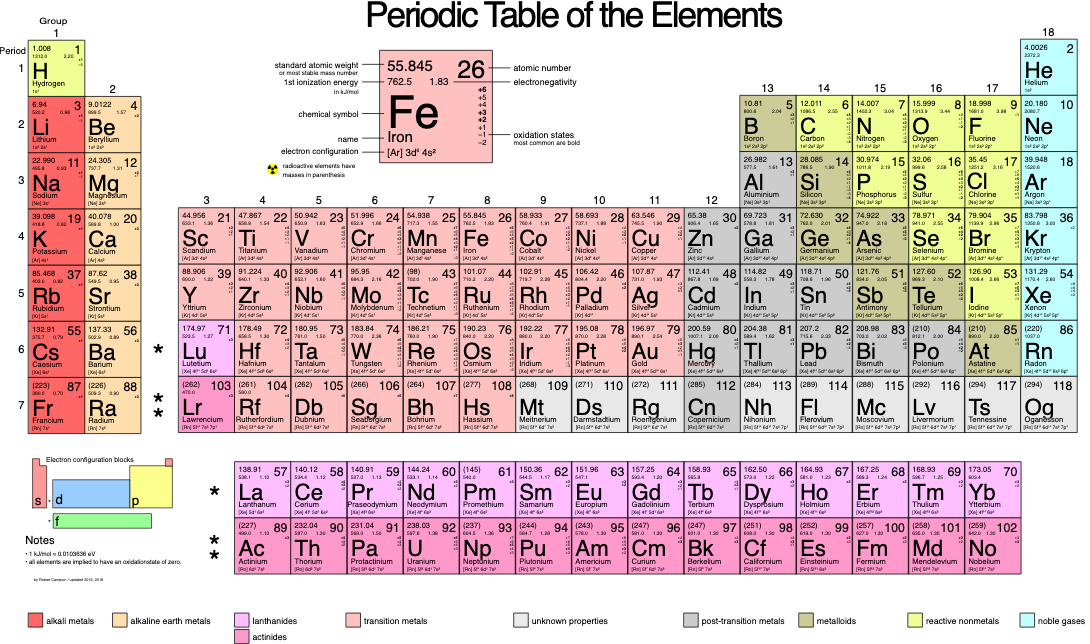
Image courtesy of Wikimedia Commons
- Identify the atomic weight. To begin, take a look at our period above and grab the atomic weight data we need. For example, F has an atomic weight of around 19 atomic mass units (amu), while Xe is around 131 amu.
- Consider the atomic ratio in the compound. , for example, may seem like a really really complicated molecule but we can break it down as a compound with 3 Cs, 52 Hs, and 12 Os with atomic weight info we already know from step 1!
- Do the math! Add up the atomic weights of all atoms present in the molecule, which would give you the molecular weight.
Now that we know the process, let’s practice by finding the molecular weight of not one but six H₂O molecules! To break it down step by step:
- Atomic weight: H’s is around 1 amu, and O’s is around 16 amu.
- Atomic ratio: In this case, we have 12 Hs and 6 Os.
- Math:
The same process applies to ionically bounded compounds; for example, let’s look at NaCl:
- Atomic weight; Na’s is around 23 amu, and Cl’s is around 35.5 amu.
- Atomic ratio: We have a 1 Na:1 Cl ratio here. Lucky for us!
- Math:
Pretty straightforward, don’t you think? 👍
Applications in Stoichiometry
Empirical formulas show the simplest whole-number ratio between elements in a compound, while molecular formulas give the exact number of each type of atom.
For example, and have different molecular formulas, but their empirical formulas are the same: !
❓ Practice Question:
Given percent composition data, calculate an empirical formula for a compound with 40% carbon, 6.7% hydrogen, and 53.3% oxygen by mass.
To solve this, we can assume that for every 100 grams of this compound, there will be 40% C, 6.7 H, and 53.3 O. Then, we can divide by their molar masses!
This makes our molecular formula ~, and this compounds empirical formula, therefore, is CH₂O.
Another application where knowing the molecular weight helps is balancing chemical equations. Although more time-consuming than simply balancing the coefficients of each molecule, we can verify that our chemical equation is balanced if each side of the equation has an equal mass by using molecular weights.
❓ Practice Question:
Is this equation balanced: ?
- Atomic weights: H = 1 amu, N = 14 amu
- Atomic ratios: 4 Ns and 4 Hs on the reactants (left) side, 1 N and 3 Hs on the products (right) side. Again, we can always stop at this step as we could already tell than the number of Ns and Hs are unbalanced, but the next step helps explain why!
- Math:
Very imbalanced, indeed!
🌐 Molar Mass, Formula Weight, and Atomic Weight Relationships
Molar Mass vs. Atomic Weight
You’ll see the words “atomic weight” and “molar mass” thrown out a lot, and it’s easy to confuse one for the other.
- The molar mass specifically refers to how much one mole of a substance weights in grams. Oftentimes, you’ll see this expressed in grams per mole (g/mol) units.
- Meanwhile, the atomic weight is specific for each element as seen on the periodic table
Why Do Molar Masses Matter?
You might wonder: “what’s the point of all this?” Understanding molar masses, as hinted in the previous study guide, helps chemists in…
- preparing solutions with accurate concentrations
- determining empirical formulas from experimental data, and
- performing stoichiometric calculations accurately during chemical reactions
💡 Guided Practice Questions
Let’s put what we’ve learned so far with these quick practice problems.
❓ Question #1: Calculate the Molecular Weight
What is the molecular weight of glucose (C₆H₁₂O₆)?
First, we identify each element’s atomic weight:
- C: ~12 amu
- H: ~1 amu
- O: ~16 amu
Math time! In this case, we have 6 Cs, 12 Hs, and 6 Os…
❓ Question #2: Converting Grams to Moles
How many moles are there in 36 grams of water?
Given the atomic weights of H and O from Example Question #1, the molecular weight of H₂O is 1 + 1 + 16 = 18 amu. In the context of molar masses, we can also say that water has a molar mass of 18 g/mol.
Using dimensional analysis, we go from grams per mole to moles with the help of molar mass:
We find that there are 2 moles of water in 36 grams of water. Woo-hoo! 🌊
Remember: this conversion factor works both ways!
For visual learners, it might be helpful to include diagrams showing how atomic weights add together within molecules or formula units for clarity. Graphics representing stoichiometry problems as pie charts or flow diagrams could also aid understanding. 🧑🎨
⭐ Wrapping Up Molecular and Formula Weights
Previously, we established what a mole is in terms of quantifying particles, atoms, and molecules. This time, we connected moles to the elements and compounds we might feel inclined to study, but to do so, we need to understand the idea of atomic and molecular weights and know how to connect them with molar masses.
By having a strong grasp on how to extract atomic weight information, we can find the molar masses of much more complex molecules and interconvert between moles, mass (typically in grams), and even number of molecules (with the help of Avogadro’s number), which’ll make life so much easier for us! 💯
5.2 Understanding Molecular and Formula Weights
1 min read•february 27, 2024
Now that we got the concept of the mole familiarized with in the previous guide, a silly question you might have when first introduced to the idea that atoms and molecules have “weight” and “masses,” too, is: “How do we even weigh them?” 😳
Good news, you’re in the right place! This guide will help you unravel the mystery of how we quantify the mass of molecules and ionic compounds, which is necessary to do calculations in chemistry.
📐 Calculating Molecular and Formula Weights
From Chemical Formulas to Weights
We briefly touched upon this when looking at mole fundamentals, but now that we’re diving into molecular and formula weights, let’s break this process down into steps to make things easier for ourselves! 😁

Image courtesy of Wikimedia Commons
- Identify the atomic weight. To begin, take a look at our period above and grab the atomic weight data we need. For example, F has an atomic weight of around 19 atomic mass units (amu), while Xe is around 131 amu.
- Consider the atomic ratio in the compound. , for example, may seem like a really really complicated molecule but we can break it down as a compound with 3 Cs, 52 Hs, and 12 Os with atomic weight info we already know from step 1!
- Do the math! Add up the atomic weights of all atoms present in the molecule, which would give you the molecular weight.
Now that we know the process, let’s practice by finding the molecular weight of not one but six H₂O molecules! To break it down step by step:
- Atomic weight: H’s is around 1 amu, and O’s is around 16 amu.
- Atomic ratio: In this case, we have 12 Hs and 6 Os.
- Math:
The same process applies to ionically bounded compounds; for example, let’s look at NaCl:
- Atomic weight; Na’s is around 23 amu, and Cl’s is around 35.5 amu.
- Atomic ratio: We have a 1 Na:1 Cl ratio here. Lucky for us!
- Math:
Pretty straightforward, don’t you think? 👍
Applications in Stoichiometry
Empirical formulas show the simplest whole-number ratio between elements in a compound, while molecular formulas give the exact number of each type of atom.
For example, and have different molecular formulas, but their empirical formulas are the same: !
❓ Practice Question:
Given percent composition data, calculate an empirical formula for a compound with 40% carbon, 6.7% hydrogen, and 53.3% oxygen by mass.
To solve this, we can assume that for every 100 grams of this compound, there will be 40% C, 6.7 H, and 53.3 O. Then, we can divide by their molar masses!
This makes our molecular formula ~, and this compounds empirical formula, therefore, is CH₂O.
Another application where knowing the molecular weight helps is balancing chemical equations. Although more time-consuming than simply balancing the coefficients of each molecule, we can verify that our chemical equation is balanced if each side of the equation has an equal mass by using molecular weights.
❓ Practice Question:
Is this equation balanced: ?
- Atomic weights: H = 1 amu, N = 14 amu
- Atomic ratios: 4 Ns and 4 Hs on the reactants (left) side, 1 N and 3 Hs on the products (right) side. Again, we can always stop at this step as we could already tell than the number of Ns and Hs are unbalanced, but the next step helps explain why!
- Math:
Very imbalanced, indeed!
🌐 Molar Mass, Formula Weight, and Atomic Weight Relationships
Molar Mass vs. Atomic Weight
You’ll see the words “atomic weight” and “molar mass” thrown out a lot, and it’s easy to confuse one for the other.
- The molar mass specifically refers to how much one mole of a substance weights in grams. Oftentimes, you’ll see this expressed in grams per mole (g/mol) units.
- Meanwhile, the atomic weight is specific for each element as seen on the periodic table
Why Do Molar Masses Matter?
You might wonder: “what’s the point of all this?” Understanding molar masses, as hinted in the previous study guide, helps chemists in…
- preparing solutions with accurate concentrations
- determining empirical formulas from experimental data, and
- performing stoichiometric calculations accurately during chemical reactions
💡 Guided Practice Questions
Let’s put what we’ve learned so far with these quick practice problems.
❓ Question #1: Calculate the Molecular Weight
What is the molecular weight of glucose (C₆H₁₂O₆)?
First, we identify each element’s atomic weight:
- C: ~12 amu
- H: ~1 amu
- O: ~16 amu
Math time! In this case, we have 6 Cs, 12 Hs, and 6 Os…
❓ Question #2: Converting Grams to Moles
How many moles are there in 36 grams of water?
Given the atomic weights of H and O from Example Question #1, the molecular weight of H₂O is 1 + 1 + 16 = 18 amu. In the context of molar masses, we can also say that water has a molar mass of 18 g/mol.
Using dimensional analysis, we go from grams per mole to moles with the help of molar mass:
We find that there are 2 moles of water in 36 grams of water. Woo-hoo! 🌊
Remember: this conversion factor works both ways!
For visual learners, it might be helpful to include diagrams showing how atomic weights add together within molecules or formula units for clarity. Graphics representing stoichiometry problems as pie charts or flow diagrams could also aid understanding. 🧑🎨
⭐ Wrapping Up Molecular and Formula Weights
Previously, we established what a mole is in terms of quantifying particles, atoms, and molecules. This time, we connected moles to the elements and compounds we might feel inclined to study, but to do so, we need to understand the idea of atomic and molecular weights and know how to connect them with molar masses.
By having a strong grasp on how to extract atomic weight information, we can find the molar masses of much more complex molecules and interconvert between moles, mass (typically in grams), and even number of molecules (with the help of Avogadro’s number), which’ll make life so much easier for us! 💯

Resources
© 2024 Fiveable Inc. All rights reserved.
AP® and SAT® are trademarks registered by the College Board, which is not affiliated with, and does not endorse this website.
